 Wholesale High Quality Walker Walking Aids to Adjustable Height for Adults
Wholesale High Quality Walker Walking Aids to Adjustable Height for Adults
 Medical Supply Foam Dressing Band Aid Bandage for Painless Dressing Change
Medical Supply Foam Dressing Band Aid Bandage for Painless Dressing Change
 Best Weight-Sensing Fall Management White Wireless Bed Sensor Pad for Hospital
Best Weight-Sensing Fall Management White Wireless Bed Sensor Pad for Hospital
 Professional 23-32mmhg Medical Varicose Calf Compression Stockings
Professional 23-32mmhg Medical Varicose Calf Compression Stockings
 Pediatric Wheelchair Near Me Motorized Wheelchair for Child
Pediatric Wheelchair Near Me Motorized Wheelchair for Child
 Intelligent Air-Operated Physiotherapy Machine Rehabilitation Robot Glove
Intelligent Air-Operated Physiotherapy Machine Rehabilitation Robot Glove
 Medical Hip Brace Orthopedic Children Hinged Hip Abduction Orthosis
Medical Hip Brace Orthopedic Children Hinged Hip Abduction Orthosis
 Hot Sale Running Knee Orthopedic Patellar Tendon Stabilizing Leg Knee Brace
Hot Sale Running Knee Orthopedic Patellar Tendon Stabilizing Leg Knee Brace
Ningbo Seaoriva Medical Co., Ltd. is a leading China-based manufacturer of high-quality medical consumables and rehabilitation products. We specialize in general disposable medical products, pharmacy products, and rehabilitation & incontinence care solutions. With years of experience, we deliver safe, reliable, and innovative medical solutions to hospitals, clinics, and pharmacies worldwide.
Every product is manufactured under strict quality control standards, ensuring patient safety, comfort, and hygiene. We leverage modern production technologies and a rigorous quality management system (ISO 13485) to maintain consistent product excellence across our comprehensive portfolio.

Guided by the principles of "quality first, innovation-driven, and customer-focused," Seaoriva Medical has earned an excellent reputation globally. Our professional R&D team and modern production facilities allow us to offer customized solutions tailored to specific clinical needs.
Analyzing the shift toward micro-needle technology and safety-engineered devices.
The industry is moving toward higher gauge needles (31G, 32G, and even 34G) to minimize tissue trauma and pain during daily injections. Thinner walls allow for better flow rates despite the smaller diameter.
Modern insulin syringes are engineered to reduce "dead space," the area where medication remains after injection. This ensures precise dosing and significantly reduces insulin waste, which is vital for high-cost analogs.
As single-use devices face environmental scrutiny, manufacturers are exploring bio-compatible polymers and recyclable packaging without compromising sterility or ETO (Ethylene Oxide) gas penetration.
How Seaoriva Medical supports large-scale healthcare supply chains.
| Procurement Category | Global Demand Driver | Seaoriva Strategic Solution |
|---|---|---|
| Public Health Tenders | Cost-efficiency & Volume reliability | Automated high-capacity production lines for economies of scale. |
| Hospital Pharmacy | Safety compliance & Sterile integrity | MDR-compliant CE certification and triple-validated sterilization. |
| Retail/Home Care | User ergonomics & Packaging clarity | Customized retail packaging with multi-lingual instructions. |
| NGO/Relief Org | Logistics resilience & Long shelf-life | Optimized shipping volume and 5-year validated sterility window. |
The transition from traditional syringes to integrated digital health companions.
Mastering the 3-bevel needle tip geometry and medical-grade silicone coating for "painless" penetration.
Integration of retractable needle mechanisms to prevent needle-stick injuries among healthcare workers (NSIs).
Exploring NFC-tagged syringes for automatic dose logging in diabetes management smartphone apps.
Seaoriva Medical understands that "Global" means "Local Compliance." We provide comprehensive technical documentation for local MoH (Ministry of Health) registrations, including stability reports, biocompatibility data, and clinical evaluation reports. Our team supports local distributors with marketing materials and staff training modules.

Expert insights into insulin syringe manufacturing and distribution.
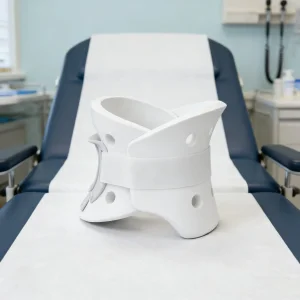 Non-Toxic Orthopedic Support Brace Safe for Sensitive Skin
Non-Toxic Orthopedic Support Brace Safe for Sensitive Skin
 Soft Cotton-Feel Disposable Adult Diaper for Men and Women
Soft Cotton-Feel Disposable Adult Diaper for Men and Women
 High Compression Stocking for Women
High Compression Stocking for Women
 Contec Tp600 Body Temperature Digital Forehead Infrared Thermometer
Contec Tp600 Body Temperature Digital Forehead Infrared Thermometer
 Wholesale Price Stainless Steel Axillary Crutch for Children
Wholesale Price Stainless Steel Axillary Crutch for Children
 Disposable Fast-Cooling Plastic Ice Pack Instant Ice Pack for Injury
Disposable Fast-Cooling Plastic Ice Pack Instant Ice Pack for Injury
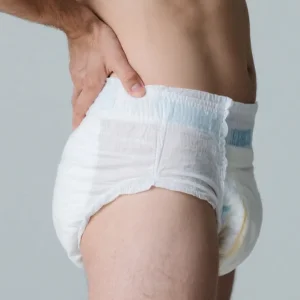 Disposable Customized Adult Diaper China Manufacture High MOQ
Disposable Customized Adult Diaper China Manufacture High MOQ
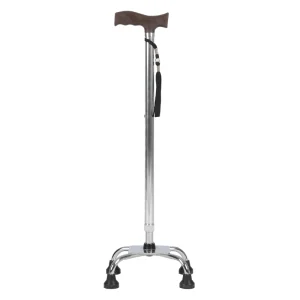 Dl-Silver 5041 Stick Walking Frame Medical Hand Cane Aluminum Crutches
Dl-Silver 5041 Stick Walking Frame Medical Hand Cane Aluminum Crutches